Accelerated and Real-Time Aging
Contact us to find out more about our global contract sterilization, testing, and validation services.
Contact UsAccelerated and Real-Time Aging Services
What is Accelerated Aging?
Accelerated aging is an artificial procedure used for establishing the shelf life to estimate the useful lifespan of a product when actual lifespan data is unavailable in an expedited manner.
Accelerated aging is conducted by simulated exposure of the product to elevated temperatures and realistic relative humidity levels for the equivalent period claimed for product expiration.
The provisional accelerated aging expiration date will remain until real time aging results are acquired.
What is Real-Time Aging?
Real-time aging is a stability testing process used to provide real data to determine the shelf life and the effects of aging on materials over time.
The packaged product is exposed to storage conditions such as temperature and relative humidity to determine its shelf life.
These studies are typically carried out at ambient temperature between 20°C to 25°C.
How is Accelerated Aging Calculated According to ASTM F1980?
Accelerated aging is calculated according to ASTM F1980 using techniques defined on the basis that the chemical reactions involved in the deterioration of materials follow the Arrhenius reaction rate.
This states that a 10°C temperature increase results in approximately two times an increase in the rate of the aging process.
The accelerated aging process is based on the relationship of temperature and reaction rate where an increase in temperature increases the reaction rate.
The level of relative humidity (RH), should be included at realistic levels.
When controlling humidity during an accelerated aging study, it is suggested in the standard to use a realistic level between 45% RH and 55% RH unless there are documented reasons for using other humidity levels.
If other levels are used, a rationale should be documented.
- It is not recommended by the standard to age packaged products at temperatures above 60°C unless knowledge of the materials shows otherwise
- The calculated duration is normally rounded up to the nearest whole day
- Ambient temperature Real Time Temperature (TRT) is typically between 20°C to 25°C
- The aging factor of 2.0 is the most common value but other levels between 1.8 – 2.5 may be used once sufficient knowledge of the materials are known and can be justified
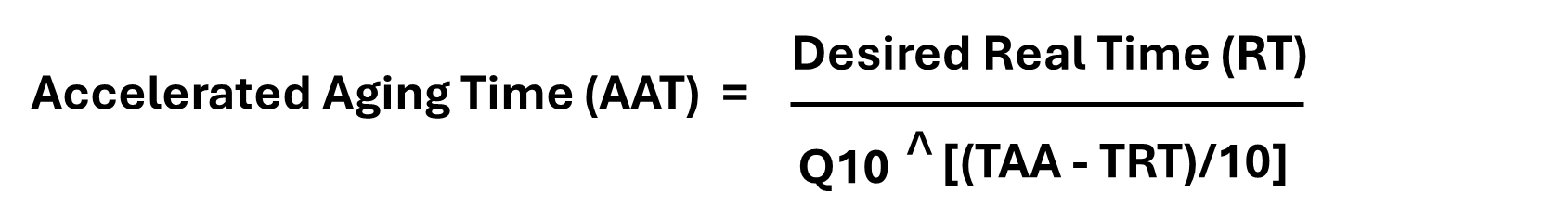
What are the Product Temperature Storage Labelling Considerations?
Where a product is labeled with specific storage temperature range, the upper temperature listed should be used when calculating the accelerated aging duration.
This is instead of the normal ambient temperature (TRT), which is typically 20°C or 25°C.
If the labelled range is 15°C to 35°C, then the 35°C should be used as the ambient temperature TRT in the calculation.
What are the Recommended Timepoints?
It is recommended that minimum of two shelf life timepoints are used.
This provides a backup if post aging tests do not meet acceptance criteria for a particular timepoint.
STERIS offers a range of accelerated aging chambers, which can accommodate both large pallets and single shipping units.
Stability Testing
What are Stability Studies?
Stability studies are conducted to evaluate the quality of a pharmaceutical or drug product over time to evaluate the influence of various environmental factors such as temperature, humidity, and light.
Typically, under accelerated conditions, this simulates the effects of long-term storage in a shorter period of time.
STERIS also offers stability testing capabilities in accordance with ICH guidelines for pharmaceutical manufacturers.
Conditions include:
- 5°C
- 25°C, 60% RH
- 30°C, 65% RH
- 30°C, 75% RH
- 40°C, 75% RH
Technical Support
Our TechTeamSM Technical Professionals also offer guidance toward:
- Packaging evaluations and concept reviews
- Risk assessment
- Test planning and protocol development
- Evaluating prior validations and gap analysis
Why Choose STERIS for Accelerated and Real-Time Aging Testing?
With over 50 years of experience as a global leader, STERIS AST provides safe and effective accelerated and real time aging with comprehensive laboratory testing and technical support through every stage of the sterilization design process - from product development to routine processing.
- Global Strength, Local Focus: Facilities across the Americas, Europe, Asia, and South Africa
- Customer Experience: Continuous feedback to drive process improvements
- Industry Leadership: TechTeamSM Technical Professional guidance through detailed and complex sterilization and laboratory testing processes
- Technology-neutral: Radiation and gas technologies tailored to Customer needs
- Excellence in Quality: Proactive approach to drive efficiencies, reduce rework, and improve quality
- Innovative Technologies: Innovative offerings to support routine services and processing redundancy capacity
- Service Commitment: Dedicated to timely routine processing and testing turn times
- Business Investments: Upgraded equipment and quality systems, new locations, and expansions of existing facilities
Related content:
TechTalk: Accelerated aging and real time studies
TechTalk: Testing validation TechTalk webinars

